Copyright 2012 neutronsources.org | All rights reserved. | Powered by FRM II | Imprint / Privacy Policy
How do protein crystals grow?
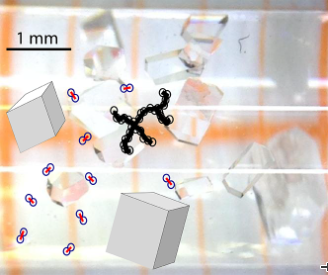
Two lysozyme molecules float as dimers in the solution, illustrated by two circles with a red connecting line. Branched fractals (black circles connected by thick black lines) coexist with the growing crystals (gray cubic objects). In the background you can see a photo of lysozyme crystals in front of a graph paper background.
How do protein crystals grow?
Researchers from MLZ (Forschungszentrum Jülich) investigated the crystallization of the protein lysozyme in an experiment at the Institut Laue-Langevin (ILL).
They focused on conditions that result in particularly large crystals, which would be sufficient for the method of high-resolution neutron protein crystallography. In neutron protein crystallography, there is the particular need for producing large crystals to obtain atomic resolution. Many applications of this method fail due to the lack of sufficiently large crystals.
The studies on the crystallization of lysozyme showed that fractals play a major role in crystal growth. Fractals are objects of individual lysozyme molecules, which are always arranged in the same structure even at infinite magnification. The lysozyme molecules initially form long straight chains. In a later stage of crystallization, the chains increasingly branch out and form a self-similar three-dimensional fractal object. These objects continue to grow and then they form the first crystal nuclei. Later, these objects are then incorporated as a whole in the growing crystals.
Previously, scientists had studied the special role of fractals using light scattering techniques only. The Jülich researchers were able to support their findings by the combination of three different measurement techniques applied to the very same sample. They used a static light scattering device and a special in-istu dynamic light scattering set-up at the same sample cell which was in the neutron beam of the small angle neutron scattering instrument D11 of the ILL. Previously, they had tested their experimental setup on the small-angle scattering instrument KWS-2 at the Heinz Maier-Leibnitz Zentrum.
Their result is a very detailed description of the crystallization of lyoszyme and allows improvements in the crystallization conditions for other proteins. This may lead to the growth of sufficiently large crystals of many other proteins for neutron protein crystallography and will therefore widen the scientific application of this method.
Crossover from a Linear to a Branched Growth Regime in the Crystallization of Lysozyme
R. J. Heigl, M. Longo, J. Stellbrink, A. Radulescu, R. Schweins, and T. E. Schrader
Crystal Growth & Design
DOI: 10.1021/acs.cgd.7b01433
Publication Date (Web): January 23, 2018