Copyright 2012 neutronsources.org | All rights reserved. | Powered by FRM II | Imprint / Privacy Policy
Neutrons could discover the key to next generation HIV drugs
Date: 12/05/2016
Source: www.ill.eu
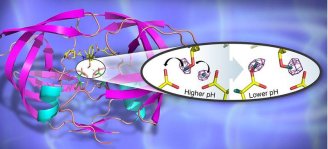
- HIV-1 protease is an enzyme which is essential for the replication of HIV, the retrovirus that causes AIDS. The enzyme is a key drug target for HIV/AIDS therapy; understanding its structure and function at the atomic level, including the location and movement of hydrogen atoms, is vital for understanding drug resistance and guiding rational drug design
- Scientists have used neutron crystallography to determine the structures of HIV-1 protease/drug complexes, providing key details of hydrogen-bonding interactions in the active-site and revealing a pH-induced proton ‘hopping’ mechanism that guides HIV-1 protease activity.
HIV, the virus that causes AIDS, has become one of the world’s most serious health and development challenges. Currently, there are approximately 36.9 million people living with HIV and tens of millions of people have died of AIDS-related causes since the beginning of the epidemic in 1981 (1). HIV not only affects the health of individuals, it impacts households, communities, and the development and economic growth of nations – there is still no cure.
HIV treatment includes medications to prevent and treat the many opportunistic infections that can occur when the immune system is compromised by HIV, as well as the use of antiretroviral therapy (ART) to attack the virus itself, with the aim of halting the development of AIDS. ART, first introduced in 1996, has led to dramatic reductions in morbidity and mortality; globally, 40% of people living with HIV are receiving treatment. HIV-1 protease is an enzyme responsible for maturation of virus particles into infectious HIV virions, which ultimately leads to the development of AIDS. Without effective HIV-1 protease activity, HIV virions remain non-infectious – with this integral role in HIV replication, the disruption of HIV-1 protease activity is therefore a key target for successful ART drugs.
The design of effective ART drugs has been led by the structures of HIV-1 protease/drug complexes determined using X-ray crystallography, and although this has led to the development of commercially available drugs, a limitation of the method is that the positions of mobile hydrogen atoms and protons cannot be determined using X-rays, and yet knowledge of their location and movement is vital for guiding the design of more effective drug therapies since hydrogen-bonding interactions play a key role in how effective a drug binds to its target.
Recently however, a collaboration between Georgia State University, USA, Oak Ridge National Laboratory, USA (ORNL), and the Institut Laue-Langevin, France (ILL) has used neutron crystallography to probe the structure of HIV-1 protease in complex with the clinical inhibitor darunavir, allowing details of the hydrogen-bonding interactions in the active site to be determined and revealing ways to enhance drug-binding and reduce drug-resistance. The group was also able to shed light on the sensitivity to pH of the enzyme’s catalytic activity.
By determining structures at different pHs, the group was able to directly observe the positions of hydrogen atoms before and after a pH-induced two-proton transfer between the drug and enzyme. The low-pH proton configuration in the catalytic site, critical for the catalytic action of this enzyme, was shown to be triggered by electrostatic effects arising from protonation state changes of surface residues far from the active site. These details can help assist in the design of new more effective ART drugs and were only possible through the use of neutron crystallography.
ILL Instrument Scientist Dr Matthew Blakeley said: “These results highlight that neutrons represent a superb probe to obtain structural details for proton transfer reactions in biological systems.”
R&D Scientist at the ORNL, Dr Andrey Kovalevsky added: “Darunavir’s structure allows it to create more hydrogen bonds with the protease active site than most drugs of its type, while the backbone of HIV-1 protease maintains its spatial conformation in the presence of mutations, meaning Darunavir-protease interaction is less likely to be disrupted by a mutation. Given these characteristics, Darunavir is an excellent therapy target to refine and therefore enhance HIV treatment.” In fact, in the US and UK, healthcare costs were estimated to be lower with Darunavir than other similar drugs, understandably making Darunavir a key focus for drug innovation in the HIV therapy area. (2)
Direct observation of proton transfer in chemical and biological systems is challenging; macromolecular neutron crystallography has been pivotal in providing key details regarding hydrogen that were required in order to answer long-standing questions about the enzyme mechanism of this important HIV drug target. Moreover, the observation that changes in amino-acid protonation-states distant from the active-site can trigger a change in hydrogen configuration in the active-site may apply to other aspartic proteases, and perhaps enzymes more generally. With the recent improvements that have been made, the field of macromolecular neutron crystallography is expanding, with studies addressing a variety of important biological processes from protein-folding to antibiotic resistance and proton transport across biological membranes.
(1) UNAIDS. How AIDS Changed Everything; 2015.
(2) McKeage K, Perry CM, Keam SJ. Drugs 2009;69(4):477-503. doi:10.2165/00003495-200969040-00007
Re.: Long-Range Electrostatics-Induced Two-Proton Transfer Captured by Neutron Crystallography in an Enzyme Catalytic Site, Gerlits et al., Angewandte Chemie International Edition 55, 4924-4927, 2016, DOI: 10.1002/ange.201509989.
Contact: Fareha Lasker, Proof communication. +44 (0)207 193 5268
Notes to Editors:
- Neutron diffraction data at ORNL were collected using the IMAGINE instrument, beam line CG-4D at the High Flux Isotope Reactor (HFIR) — a DOE Office of Science User Facility. UT-Battelle manages ORNL for the DOE’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit http://science.energy.gov/. Support for the preparation of deuterated HIV-1 protease was provided by the Center for Structural Molecular Biology (CSMB), DOE Office of Biological and Environmental Research (BER).
- Neutron diffraction data at ILL were obtained using the LADI-III instrument. LADI-III uses a large cylindrical area detector composed of neutron-sensitive image-plates, which completely surround the crystal and allows large numbers of reflections to be recorded simultaneously. Data are collected using a quasi-Laue method in order to provide a rapid survey of reciprocal space, while reducing the background on the detector compared to use of the full white beam. Data collection is feasible for samples with unit-cell edges ranging from ~50 to 150 Å using crystal volumes from ~0.05 to 0.5mm3, respectively. For more information please visit https://www.ill.eu/
About ILL – the Institut Laue-Langevin (ILL) is an international research centre based in Grenoble, France. It has led the world in neutron-scattering science and technology for almost 40 years, since experiments began in 1972. ILL operates one of the most intense neutron sources in the world, feeding beams of neutrons to a suite of 40 high-performance instruments that are constantly upgraded. Each year 1,200 researchers from over 40 countries visit ILL to conduct research into condensed matter physics, (green) chemistry, biology, nuclear physics, and materials science. The UK, along with France and Germany is an associate and major funder of the ILL.
About ORNL – Oak Ridge National Laboratory (ORNL) operates two neutron source facilities, the High Flux Isotope Reactor and the Spallation Neutron Source. Built and funded by the U.S. Department of Energy (DOE) Office of Basic Energy Sciences (BES), the two facilities combined house 30 neutron scattering instruments, providing researchers with unmatched capabilities for understanding the structure and properties of materials, macromolecular and biological systems, and the fundamental physics of the neutron. More than 2800 researchers from around the world use ORNL’s neutron sources annually. ORNL is managed and operated by UT-Battelle for DOE.