Copyright 2012 neutronsources.org | All rights reserved. | Powered by FRM II | Imprint / Privacy Policy
Neutrons probe biological materials for insights into COVID-19 virus infection
Date: 18 September 2020
Source: ORNL
SARS-CoV-2, the coronavirus responsible for the disease COVID-19, is infecting the world at a rapid rate. Understanding how this infection works at the molecular level could help experts discover ways to moderate or stop the spread.
A team of scientists at the Department of Energy’s (DOE’s) Oak Ridge National Laboratory (ORNL) is using neutron reflectometry to do just that.
Neutrons are capable of probing biological materials under physiological conditions without damaging them. By harnessing these properties, the researchers can measure the infection dynamics of the virus as it happens.
Their mission is to get a detailed look at some of the first stages of infection occurring at the cell membrane. These findings will help the team test antiviral drug candidates that could disrupt this process. The data obtained from these experiments could also inform other studies focused on developing therapeutics and vaccines.
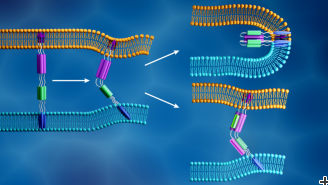
The researchers are focusing their analysis on SARS-CoV-2 spike proteins, barb-like structural proteins that cover the surface of the virus and trigger the infection process. The spike protein binds to a receptor on the host cell’s outer layer and facilitates fusion between the viral and cellular membranes, allowing the virus to enter the cell and release its genetic material. The cell’s protein-making machinery then uses this genetic information to make new copies of the virus.
When SARS-CoV-2 hijacks a host cell, its spike protein splits into two subunits, called S1 and S2. The two parts are both essential for infection. The S1 subunit contains a receptor binding domain that recognizes and latches on to a cell receptor. Cell receptors are proteins embedded within the cell membrane that can bind to specific molecules outside the cell. This connection can cause the components to change shape, which in turn could induce cascading changes within the cell. For the SARS-CoV-2 spike protein, this connection activates the S2 subunit, which helps the virus merge its membrane with that of the cell. Therefore, the spike protein’s function is similar to opening a locked door, with S1 as the key that unlocks the door and S2 as the force that pushes the door open.
Learning from past epidemics
The overall structure of the SARS-CoV-2 spike protein is very similar to that of SARS-CoV, an earlier coronavirus that caused severe acute respiratory syndrome (SARS), and this resemblance helped the team develop their research strategy.
The S1 subunit is the focus of many drug development studies, since this part of the spike protein has been shown to provoke an immune response in the human body. However, previous SARS-CoV studies found the S1 subunit experiences high rates of mutation, allowing the virus to evade antibody-based treatments while maintaining its ability to infect cells. “This is the lesson we learned from the original SARS epidemic,” said Minh Phan, a postdoctoral research associate at ORNL and principal investigator of this project.
Phan and his colleagues are studying the S2 subunit because this component of the spike protein does not mutate as quickly. Treatments that prove successful in inhibiting the S2 function may stay effective for a longer time.
A nanoscale view of coronavirus
To better understand the dynamics of viral S2 subunits and host cell membranes, the researchers are employing the liquids reflectometer (LIQREF) at ORNL’s Spallation Neutron Source (SNS). By measuring how neutrons reflect at different angles when passing through different types of matter, the instrument can help shed light on the structure of biological materials at the molecular scale.
The team first synthesized a lipid membrane that mimics the outer membrane of cells that line the surfaces inside human lungs, where this viral infection can take place. They identified how the lipids were organized within the membrane and how this arrangement changes when the membranes are exposed to different conditions, such as temperature, pressure, and acidity.
At the LIQREF instrument, the researchers spread the lipid membrane atop a thin layer of water in an apparatus called a Langmuir trough. They then introduce the S2 subunit to these membranes to observe in detail how the S2 and lipid membranes change shape when they interact.
Neutrons are also ideal for this study because they are sensitive to the element hydrogen, common to all biological molecules, and its isotopes. By replacing some hydrogen atoms with deuterium atoms, scientists can create contrast in their samples and selectively zero in on different structural features. This technique is useful for studying samples that involve multiple components with similar densities, like lipid membranes.
“Generally, these membranes are not single-lipid membranes,” said John Ankner, an instrument scientist involved with this study. “They consist of lipids of a certain structure, lipids of another structure, cholesterol, proteins, and things that come in contact with them.”
To capture this complexity, the research team is investigating multiple versions of the membrane, changing the contrast of the sample with deuterium each time.
“By taking multiple measurements and assembling all of this information together, you can create a single picture of how these different components go together,” said Ankner.
The information derived from these experiments will then help steer the team’s efforts in selecting and testing drug candidates that could block this interaction, such as fusion inhibitors that successfully blocked original SARS-CoV infections. If these inhibitors can stop the new coronavirus from invading healthy cells, existing drugs could potentially be repurposed to treat COVID-19 patients. The results may also help guide the design of new fusion inhibitors.
Capturing infection
While other studies have used protein crystallography to better understand the atomic structure of the coronavirus S2 subunit alone, this project is analyzing how S2 changes shape when interacting with a lipid membrane. A shape change could be important for inducing actions within a cell after the spike S1 subunit binds to the cell receptor. Phan also notes that the LIQREF instrument allows the team to measure these dynamics under physiological conditions, whereas protein crystallography only allows researchers to capture what the S2 subunit looks like in a crystallized form.
“At ORNL, we have the right tools to study the dynamics of the interaction under physiological conditions. This allows us to better understand how the S2 subunit moves and changes shape naturally in a wet environment,” said Phan. “Such information could complement what experts already know about the protein from crystallography. If we can help verify what this mechanism looks like, then we may have a clearer understanding to guide the development of drugs that block the fusion process.
Collaboration is key
Of course, learning more about the S2 subunit and its certain behaviors depends on the ability to grow quality samples, which involves synthesizing S2 subunit proteins, purifying them, and preparing them for experimentation.
Phan and Ankner note that this part of their research has been made possible only through collaboration with labs across ORNL and at outside institutions.
The S2 subunit protein was synthesized in mammalian cell cultures by Steve Foster, a biomedical researcher at the University of Tennessee Medical Center in Knoxville, Tennessee. Through this method, he can develop S2 proteins for research that retain several aspects of its natural structure and function.
“In our lab we routinely use mammalian cell cultures for protein production, so we hope we’ve produced an S2 protein best suited for this research analysis. Our proximity to ORNL also works well in that the sample doesn’t have to travel far, meaning less risk of damaging the protein or distorting its original structure, which is critical for this work,” said Foster.
Following its synthesis, the sample was purified by Jessy Labbé and Michael Melesse Vergara from ORNL’s Biosciences Division. Scientists from the ORNL Neutron Sciences Directorate then performed a series of tests to confirm the structure of the sample protein and check its purity. This effort was implemented by Yichong Fan and Wellington Leite from the Bio-Labs team, and Jacob Kinnun and Mary Odom from the SNS team.
“We put an enormous effort into making sure the protein has the right properties going into the experiment. If it does not, we could get spurious results and misinterpret what we’re doing,” said Hugh O’Neill, director of ORNL’s Center for Structural and Molecular Biology and lead researcher for the Bio-Labs team.
“This virus is extremely delicate in its components, and it’s a big challenge to get these materials to the neutron instrument,” said Ankner. “That’s why involving various ORNL labs and the University of Tennessee is so crucial. Each step that eventually gets the sample onto our instrument requires the expertise of lots of people.”
This project also relied on efforts from the LIQREF instrument staff, who were instrumental in developing the systems, protocols, and modeling frameworks necessary to run the experiments and interpret the data.
“Experts across the division, across ORNL, and from partner institutes have come together for this project,” said Phan. “We couldn’t have done this without their support, and it’s greater motivation to fulfill our mission.”