Related News
Copyright 2012 neutronsources.org | All rights reserved. | Powered by FRM II | Imprint / Privacy Policy
Biology and neutrons collide to unlock secrets of fish ear bones
Scientific discovery can come from anywhere, but few researchers can say the answers to their questions would come from the pea-sized bones in the head of a six-foot-long, 200-pound prehistoric freshwater fish.
Date: 05/01/2017
Source: ornl.gov

In a unique pairing of biology and neutron science, researchers from the Department of Energy’s Oak Ridge National Laboratory have gained new insights into aquatic biochemistry using the otoliths of the lake sturgeon, Acipenser fulvescens.
Otoliths are small ear bones in fish used for hearing and balance, composed of polymorphs, or forms, of calcium carbonate called calcite, aragonite and vaterite.
Vaterite is the rarest and least stable of the polymorphs, yet is a highly sought-after biomaterial as an additive in paper, plastics, cosmetics and biomedical products such as drug-delivering nanocapsules. Despite this widespread interest, vaterite remains a mysterious substance: Researchers have proposed more than a dozen models of its poorly understood crystalline structure.
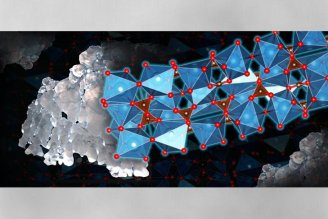
Most fish otoliths are made of aragonite, but some primitive fish species, namely sturgeons, have vaterite otoliths. Previous studies of sturgeon otoliths reported calcite fractions, or content, but were either dismissed as mistakes or as byproducts of preservation, as it was assumed the otoliths could only be pure vaterite.
Brenda Pracheil, an aquatic ecologist in ORNL’s Environmental Sciences Division, partnered with Bryan Chakoumakos, a neutron scientist in the lab’s Quantum Condensed Matter Division, to take a deeper look into sturgeon otoliths with a novel technology rarely seen in aquatic biology.
Using neutron diffraction, the pair proved the otoliths contained both vaterite and calcite fractions and validated a crystalline structural model of vaterite to advance understanding of the rare polymorph.
“We’re applying materials science techniques to studying otoliths,” Chakoumakos said. “We’re trying to add a little rigor and introduce new techniques in this emerging research area.”
Despite its high resolution and ease of use, neutron diffraction had never been used to examine the polymorph composition of otoliths. It is nearly impossible to distinguish between polymorphs by sight, and techniques such as Raman spectroscopy only sample the surface of the otolith. X-ray diffraction can find the average polymorph composition, but requires the sample be ground into powder, destroying the natural crystal orientation and integrity of the otolith.
“The nice thing about neutrons is that we’re able to easily and nondestructively get a snapshot of the whole otolith and preserve it for other measurements,” Chakoumakos said.
Carbon and oxygen atoms also scatter neutrons stronger than X-rays, allowing the team to examine the carbonate group of vaterite with greater clarity. Their data best fit a structural model corroborated by X-ray diffraction experiments, narrowing the field of proposed structures down to one reliable model.
The otolith study underscores the potential of novel collaborations among research teams with compatible scientific goals.
“It’s a pretty good collaboration because I didn’t know anything about fish other than I like to catch them with my fly rod,” Chakoumakos said. “I had casually done some neutron diffraction on otoliths I had collected. I knew there had been reports that some were vaterite and I wanted to study that material because the structure was unknown.”
Chakoumakos heard about Pracheil’s work on otolith microchemistry and contacted her with an idea to study the vaterite in sturgeon otoliths with neutron diffraction. Since then, their work has capitalized on Pracheil’s expertise in sturgeon otoliths and Chakoumakos’ experience with the instruments at the Spallation Neutron Source and High Flux Isotope Reactor, which are DOE Office of Science User Facilities.
“There hasn’t been much collaboration between environmental and neutron sciences, but there are a lot of applications to what we’re doing,” Pracheil said. “There are so many new tools all the time, but they don’t mean anything if you don’t know how they will answer your research questions.”
The next step for the team is supplementing their neutron experiments with electron backscattering diffraction and X-ray microfluorescence to generate spatial maps to better understand how differences in polymorph composition influence trace element distribution in otoliths.
“This is really revolutionary to the field of microchemistry because it says we need to consider these polymorphs as not just something trivial,” Pracheil said. “There’s a lot there and we’re just scratching the surface.”
After gaining so much knowledge about the miniscule otoliths through these novel techniques, the team can see even bigger questions in aquatic ecology, fisheries management and evolutionary biology for other scientists to explore.
“I think it’s really cool, as a biologist, that we were able to take this weird prehistoric fish and validate models and empirically describe this previously unknown crystal structure with novel techniques,” Pracheil said. “It opened my eyes to how important these materials science techniques are to our fundamental work.”
The team’s recent study in Scientific Reports, titled “Empirically testing vaterite structural models using neutron diffraction and thermal analysis,” can be found here. Former ORNL researcher Mikhail Feygenson, now with Forschungszentrum Jülich and the European Spallation Source, contributed to the study, as well as Ryan Koenigs and Ronald Bruch of the Wisconsin Department of Natural Resources. The team’s earlier study, “Sturgeon and paddlefish (Acipenseridae) saggital otoliths are composed of the calcium carbonate polymorphs vaterite and calcite,” in the Journal of Fish Biology can be found here. Mikhail Feygenson, Ryan Koenigs and Ronald Bruch contributed, as well as Gregory Whitledge of the Center for Fisheries, Aquaculture and Aquatic Sciences at Southern Illinois University.
UT-Battelle manages ORNL for the Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.
Media Contact:
Sean Simoneau, Communications
simoneausm@ornl.gov, 865.241.0709


